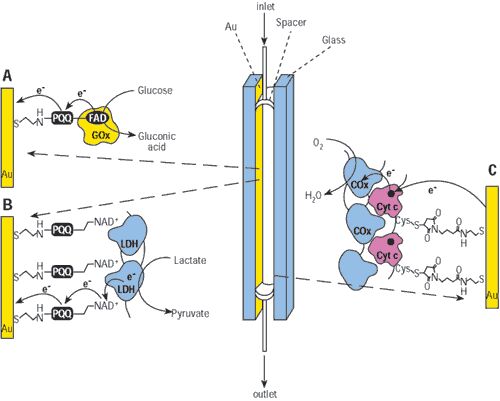
Figure: Self-powered biofuel cell biosensors with anodes that detect either (A) glucose or (B) lactate. Both sensors use (C) the cytochrome c/cytochrome oxidase - functionalized cathode.
Although most fuel cell
research is aimed at high
energy and efficiency,
Itamar Willner and his co-workers at the Hebrew University of Jerusalem
(Israel) took exactly the opposite tack. Low efficiency for their
biofuel
cells translates into highly specific information about the
concentration
of an analyte. These self-powered biosensors put out enough energy (1
µW)
for a good signal but not enough to cause contaminating reactions that
would clutter their data (see Figure).
 |
Figure: Self-powered biofuel cell biosensors with anodes that detect either (A) glucose or (B) lactate. Both sensors use (C) the cytochrome c/cytochrome oxidase - functionalized cathode. |
The sensor is essentially a flow cell made from two electrodes separated by a spacer. The anode is functionalized with an enzyme layer—in one version of the biosensor, glucose oxidase. To form an effective electrical contact between the glucose oxidase and the electrode, the researchers use a previously reported process in which the apoenzyme is reconstituted on a flavine adenine dinucleotide layer, which is covalently linked to the electrode. The enzyme catalyzes the oxidation of glucose—the analyte or fuel—to gluconic acid. This yields low-level electrical power, and the open-circuit voltage of the system is determined by the fuel concentration. The cycle is complete with the simultaneous reduction of oxygen to water on a cathode functionalized with cross-linked cytochrome c/cytochrome oxidase.
A second version of the sensor replaces glucose oxidase with lactate dehydrogenase using a similar functionalization process. The sensors could detect glucose and lactate, respectively, in the 1- to 80-mM range. The cells were operated continuously for 5–7 h. Because the cells are self-powered by biological fluids, Willner and colleagues suggest that they may be used as invasive sensing devices. The researchers also say that the sensors can be tailored to detect substances such as alcohols, fructose, and amino acids. (J. Am. Chem. Soc. 2001, 123, 10,752–10,753).
